Pharmaceutical Manufacturing Solutions
Confident decisions. Compliant processes. Reliable performance.
Pharmaceutical manufacturing operates under constant regulatory oversight while facing increasing pressure to improve efficiency, reduce variability, and maintain supply continuity.
Minitab supports pharmaceutical teams with solutions that strengthen quality systems, improve process performance, and bring consistency to data-driven decisions from development through commercial production.

The best quality products are designed with Minitab



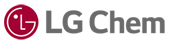


Improving Quality and Performance in Pharmaceutical Manufacturing
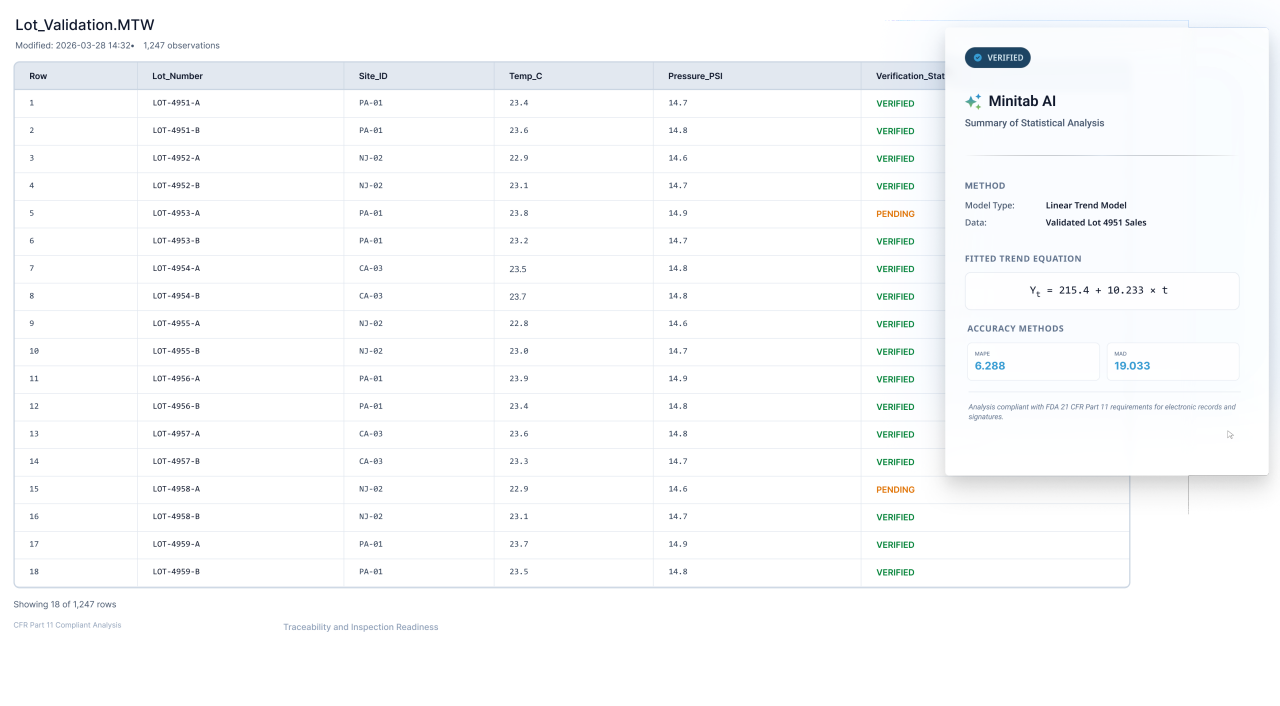
Pharmaceutical organizations are dealing with shifting regulatory expectations, global production networks, and more data than ever before. Inspection readiness is no longer periodic, it is continuous. Process validation does not end at launch, it requires ongoing performance verification and continued process monitoring across commercial manufacturing.
Yet many teams still rely on manual analysis, spreadsheets, or disconnected systems that make it difficult to maintain consistency across sites and product lines. .
To improve performance without increasing risk, pharmaceutical manufacturers need structured, repeatable approaches to analyzing data, solving problems, and monitoring processes throughout the product lifecycle.
For more than 50 years, Minitab has supported regulated industries with solutions that bring clarity, structure, and confidence to critical decisions.
Reduce experimental runs by up to 40%
Bring structure to formulation and process development using Design of Experiments (DOE) to understand variable interactions and identify critical process drivers. By replacing one-factor-at-a-time testing with structured experimentation, teams can reduce experimental runs by up to 40% while strengthening process understanding and validation readiness.
Strengthen measurement and data reliability
Ensure the data behind your release, validation, and monitoring decisions is reliable and defensible. Minitab solutions help teams evaluate measurement systems, reduce uncertainty, and build confidence in the integrity of their results.
Standardize analysis across teams and sites
Replace inconsistent spreadsheets and disconnected approaches with standardized workflows that promote repeatability, transparency, and traceability. Teams can generate consistent outputs that support internal alignment and external inspection readiness.

Read Our Guide: The FDA’s Updated Expectations for Software in Pharmaceutical Manufacturing: What They Mean and How to Respond
In this guide, we break down the FDA’s 2025 Computer Software Assurance guidance and its shift to risk based, proportionate validation for software. It also shows how manufacturers can leverage supplier validation and analytics tools like Minitab to maintain compliant, defensible quality systems.
How Pharmaceutical Teams Improve Process Performance
- 1. Import and connect your data
- 2. Apply Structured Analysis and Problem Solving
- 3. Design and Optimize Processes
- 4. Make Confident, Defensible Decisions
Pharmaceutical data exists across manufacturing equipment, laboratory systems, spreadsheets, enterprise platforms, and quality systems. Bringing this information together is the first step toward meaningful improvement.
Minitab solutions integrate with production and quality data sources, enabling teams to analyze process performance, monitor critical parameters, and identify trends without relying on fragmented manual workflows. This creates a foundation for consistent analysis across sites and departments.
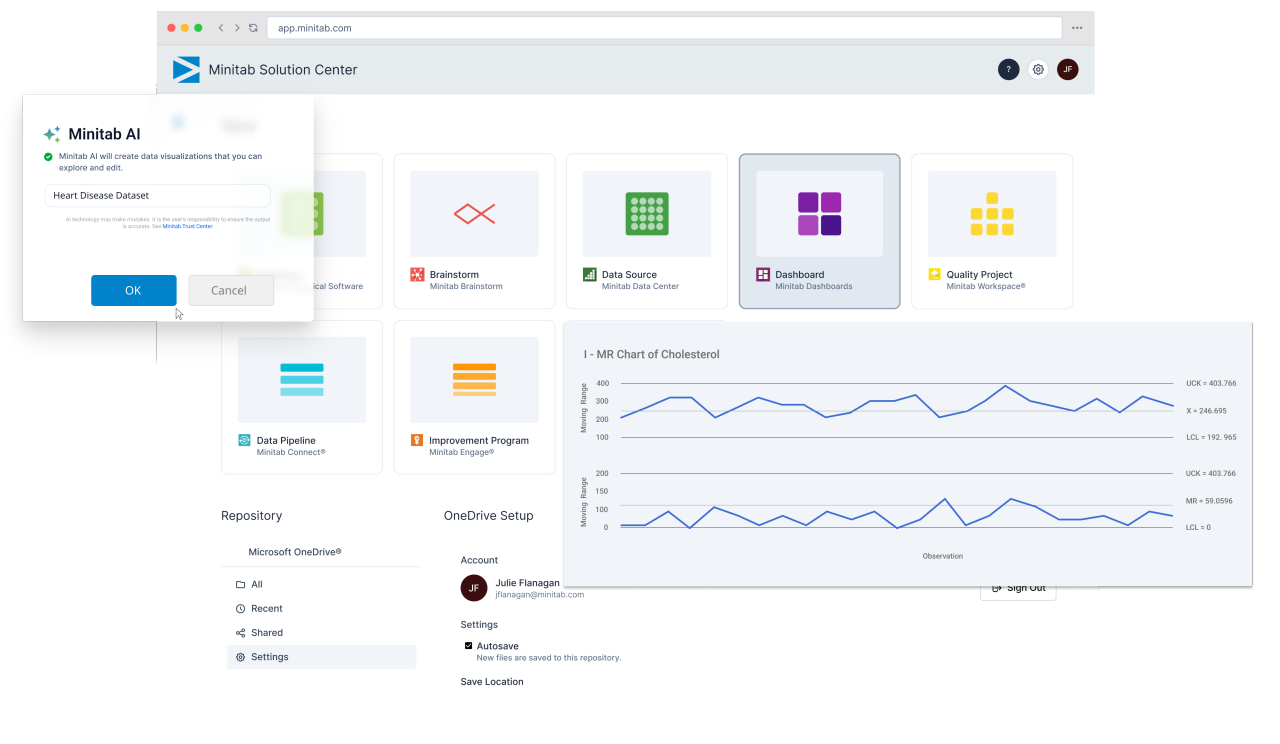
Once data is accessible, structured analysis ensures decisions are consistent and defensible. Pharmaceutical teams use Minitab solutions to support process validation activities, ongoing performance monitoring, root cause investigations, and continuous improvement initiatives.
From development through commercial production, teams can model processes, simulate outcomes, evaluate capability, and apply structured problem-solving frameworks that reduce variability and strengthen compliance.
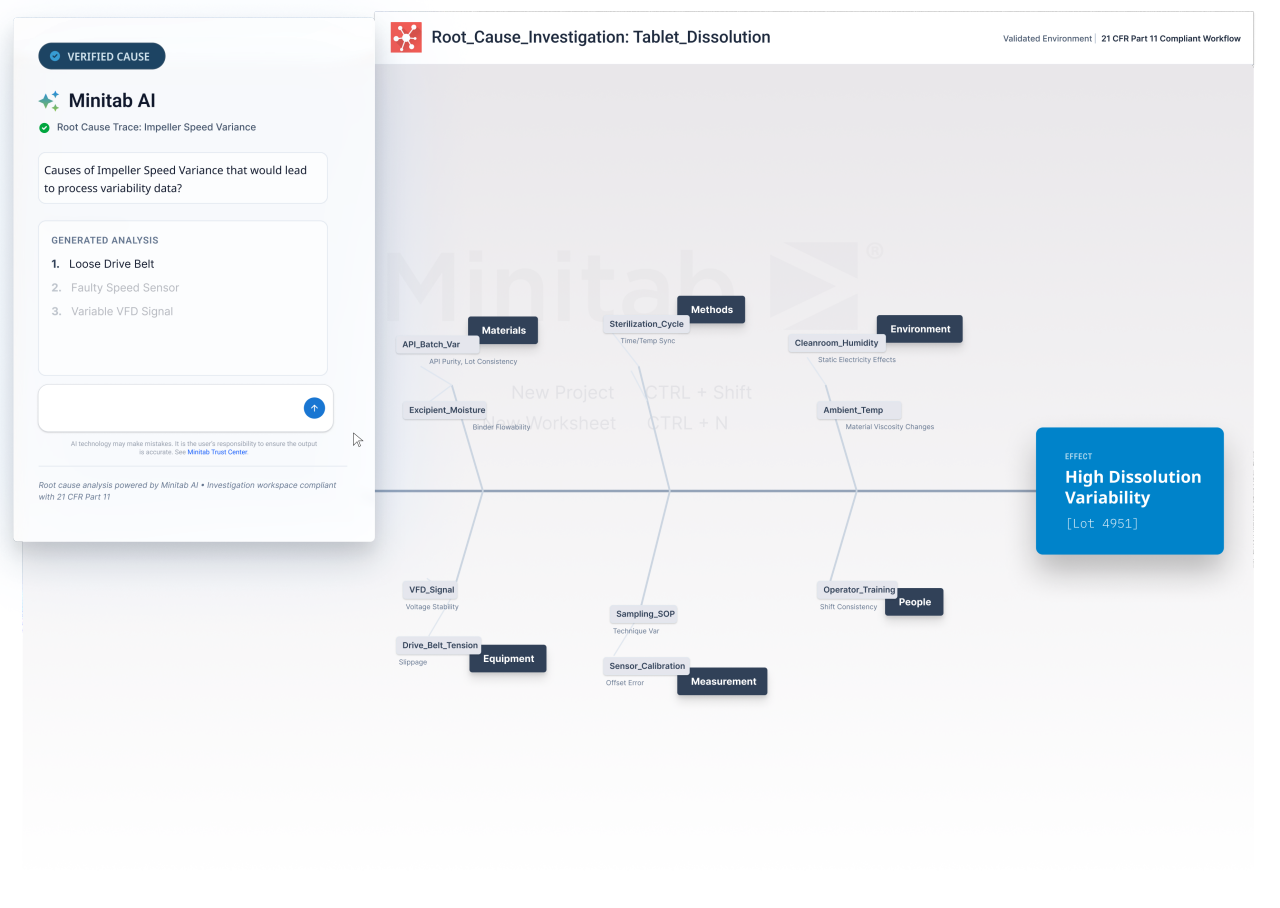
Accelerate pharma development with DOE to reduce trial and error, align teams on data driven decisions, and scale optimized processes with greater speed and confidence.
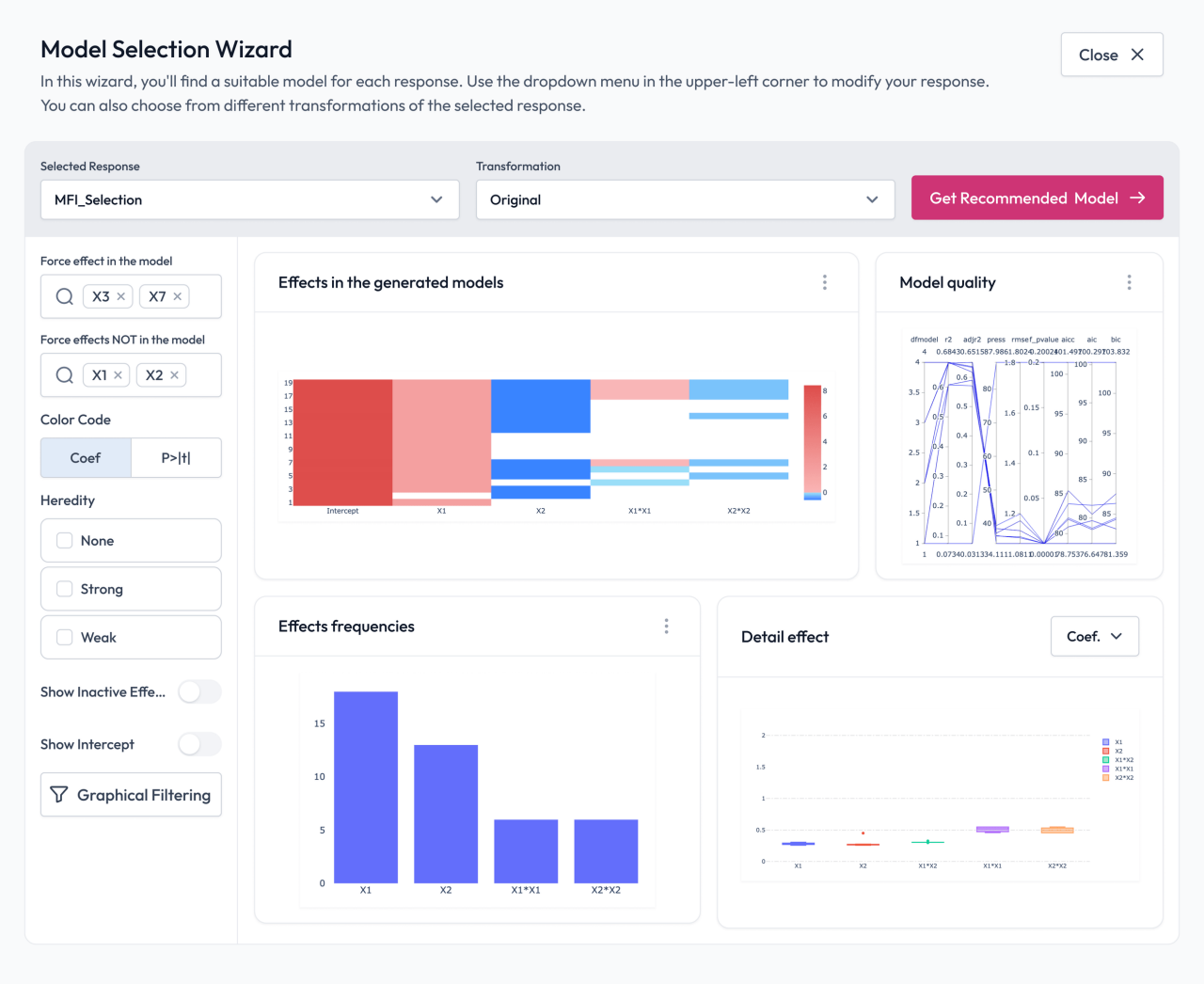
Regulatory environments demand transparency and repeatability. Pharmaceutical organizations must be able to explain how conclusions were reached and demonstrate that processes remain in control.
Minitab supports standardized analytical workflows, documentation, and reporting that promote traceability and cross-site consistency. By replacing ad hoc analysis with governed, repeatable methods, teams improve both operational performance and inspection readiness.
Ranked #17 in G2's 2026 Best Software Awards for a Reason
WHAT OUR CUSTOMERS SAY
“Minitab [is] the best tool for quality management…time series plots, control charts, Pareto charts, fishbone charts—everything presented to upper management using Minitab. I would recommend it without hesitation!”
Jose Luis P.
Quality Chief
“Using Real Time SPC and Prolink with EWMA charts, we identified temperature effects on production and enabled timely adjustments to maintain consistency.”
George
Senior Quality Process Engineer
“Minitab helps me understand failure points and relationships within complex processes. The FMEA tool alone has saved us hours of discussions and calculations.
Our efficiency has gone up significantly.”
Rahul V.
Senior Development Engineer
Trusted by pharmaceutical manufacturers worldwide
For more than 50 years, pharmaceutical manufacturers have trusted Minitab to help them improve quality, reduce risk, and strengthen operational performance. Our solutions are used by quality engineers, validation specialists, manufacturing leaders, and continuous improvement teams worldwide.
Minitab enables organizations to validate and monitor processes with confidence, strengthen data integrity, standardize problem-solving approaches, and accelerate investigations without increasing complexity. By connecting advanced analytics, Design of Experiments (DOE), real-time monitoring, simulation, and structured improvement tools, we help pharmaceutical teams reduce recurring investigations and stabilize performance before issues escalate.
Start solving bigger problems today.
Free resources engineers rely on to learn, improve, and make smarter decisions:




