Treinamento de aparelhos médicos
Treinamento presencial ou virtual

- Treinamento de aparelhos médicos
- Fundamentos do Minitab para aparelhos médicos
- Qualidade estatística para aparelhos médicos
- Experimentos fatoriais (DOE) para aparelhos médicos
- Workshop
Nossa série de “Aparelhos médicos” é para profissionais que trabalham nesse setor. Os materiais do curso incluem exemplos com métricas, como resistência à quebra, diâmetro, tamanho de partícula, não conformidades e umidade.

FUNDAMENTOS DO MINITAB PARA APARELHOS MÉDICOS
Nesse curso fundamental de 2 dias, você aprenderá a minimizar o tempo necessário para análise de dados usando o Minitab para importar dados, desenvolver abordagens estatísticas sólidas para explorar dados, criar e interpretar gráficos atraentes e exportar resultados. Analise uma variedade de conjuntos de dados reais de aparelhos médicos para aprender como alinhar suas aplicações com as ferramentas estatísticas certas e interpretar resultados estatísticos para revelar problemas com um processo ou evidências de melhoria. Aprenda os fundamentos de conceitos estatísticos importantes, como teste de hipóteses e intervalos de confiança e como revelar e descrever relações entre variáveis com ferramentas de modelagem estatística.
Esse curso enfatiza muito a tomada de decisões sólidas baseando-se na aplicação prática de técnicas estatísticas comumente encontradas no setor de aparelhos médicos.
Os tópicos incluem:
- Importação e formatação de dados
- Gráficos de barras
- Histogramas
- Boxplots
- Gráficos de Pareto
- Gráficos de dispersão
- Tabelas e análises de chi-quadrado
- Medições de local e variação
- Testes-t
- Testes de proporções
- Teste de igualdade de variâncias
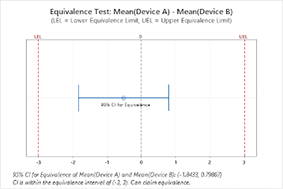
- Testes de equivalência
- Poder e tamanho de amostra
- Correlação
- Regressão linear simples e múltipla
- ANOVA com um fator
- ANOVA com várias variáveis
Pré-requisito: nenhum
QUALIDADE ESTATÍSTICA PARA APARELHOS MÉDICOS
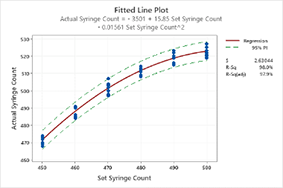
Desenvolva as habilidades necessárias para avaliar e certificar sistemas de medição. Conheça os fundamentos do controle de processo estatístico e como essas importantes ferramentas da qualidade podem fornecer a evidência necessária para aprimorar e controlar processos de aparelhos médicos. Desenvolva as habilidades necessárias para saber quando e onde usar os vários tipos de gráficos de controle disponíveis no Minitab para seus próprios processos. Aprenda a usar ferramentas importantes de análise de capacidade para validar seus processos relativos a especificações internas e do cliente.
A ênfase do curso está no ensino das ferramentas de qualidade e sua relação com os processos de aparelhos médicos.
Os tópicos incluem:
- R&R da medição
- Teste destrutivo
- Vício e linearidade de medição
- Concordância por atributos
- Variáveis e cartas de controle de atributos
- Análise de capacidade para dados normais, não normais e de atributos
- Amostragem de aceitação
Pré-requisitos: Ferramentas estatísticas para aparelhos médicos - Fundamentos do Minitab

EXPERIMENTOS FATORIAIS (DOE) PARA APARELHOS MÉDICOS
Aprenda a gerar vários experimentos fatoriais fracionais e completos usando a interface intuitiva de planejamento de experimento do Minitab. Aplicações reais de aparelhos médicos demonstram como os conceitos de aleatorização, replicação e bloqueio formam a base das práticas de experimentação seguras. Desenvolva as habilidades necessárias para analisar corretamente os dados resultantes e atingir seus objetivos experimentais de forma eficiente e eficaz.
Use os poderosos gráficos personalizáveis do Minitab para interpretar e comunicar resultados experimentais e aprimorar produtos e processos, encontrar fatores críticos que causam impacto em variáveis de resposta importantes, reduzir variação do processo e gerar projetos de pesquisa e desenvolvimento.
Os tópicos incluem:
- Planejamento de experimentos fatoriais
- Gráficos de efeitos normal e Pareto de efeitos
- Poder e tamanho de amostra
- Gráficos de efeito principal, interação e cubo
- Pontos centrais
- Gráficos de contorno sobreposto
- Otimização de respostas múltiplas
Pré-requisitos: Ferramentas estatísticas para aparelhos médicos - Fundamentos do Minitab
WORKSHOP
O treinamento do Minitab fornece a base para melhorar sua eficiência no uso de estatística para analisar dados. Os exemplos apresentam cenários do mundo real para aprender as ferramentas, enquanto os exercícios permitem tempo para praticar. Reforce o treinamento usando dados da sua empresa para concluir sua jornada educacional. Isso oferece aos participantes a oportunidade de se relacionarem diretamente com seus próprios casos de uso.
O workshop tem grande ênfase na tomada de decisões sólidas com base na aplicação prática de ferramentas estatísticas nos projetos de sua empresa usando seus dados.
Os tópicos serão determinados pelos dados específicos do cliente apresentados no workshop.
Cursos de treinamento
Entre em contato conosco se tiver alguma dúvida sobre quais cursos são adequados para você ou para agendar treinamento.